Purcell, Chemistry and Chemical Reactivity, 2nd ed., 1991. Table 2.1.3: Properties of Selected Isotopes Element (Note: we will discuss the derivation of the atomic mass in the next section). 
The properties of some common isotopes are in Table 2.1.3. Many elements other than carbon have more than one stable isotope tin, for example, has 10 isotopes. The nucleus of 14C is not stable, however, but undergoes a slow radioactive decay that is the basis of the carbon-14 dating technique used in archeology. In addition to 12C, a typical sample of carbon contains 1.11% 13C, with 7 neutrons and 6 protons, and a trace 14C, with 8 neutrons and 6 protons. Consequently, it is more often written as 12C, which is read as “carbon-12.” Nevertheless, the value of Z is commonly included in the notation for nuclear reactions because these reactions involve changes in Z.įigure 2.12: Formalism used for identifying specific nuclide (any particular kind of nucleus)Īlthough carbon-12 is the most abundant type of isotope in carbon, it is not the only isotope. The subscript indicating the atomic number is actually redundant because the atomic symbol already uniquely specifies Z. The isotope of carbon that has 6 neutrons is therefore 12 6C. An isotope of any element can be uniquely represented as A ZX, where X is the atomic symbol of the element, A is the mass number and Z is the atomic number. In a typical sample of carbon-containing material, 98.89% of the carbon atoms also contain 6 neutrons, so each has a mass number of 12. The element carbon (C) has an atomic number of 6, which means that all neutral carbon atoms contain 6 protons and 6 electrons. Mass Number(A) = Number of Protons + Number of Neutrons Because different isotopes of the same element haves different number of neutrons, each of these isotopes will have a different mass number(A), which is the sum of the number of protons and the number of neutrons in the nucleus of an atom. All isotopes of an element have the same number of protons and electrons, which means they exhibit the same chemistry. Atoms that have the same number of protons, and hence the same atomic number, but different numbers of neutrons are called isotopes. Where more than one isotope exists, the value given is the abundance weighted average. This is approximately the sum of the number of protons and neutrons in the nucleus. Relative atomic mass The mass of an atom relative to that of carbon-12. Unlike protons, the number of neutrons is not absolutely fixed for most elements. Density is the mass of a substance that would fill 1 cm 3 at room temperature. Recall that the nuclei of most atoms contain neutrons as well as protons. Hence, the atomic number defines the element in question. If you change the atomic number to 12, you are no longer dealing with sodium atoms, but magnesium atoms. That means that all sodium atoms have 11 protons. For example, the atomic number (z) for sodium (Na) is 11. The symbol for the atomic number is designated with the letter Z. This number is known as the atomic number, which identifies the number of protons in the nucleus of ALL atoms in a given element. When you study the periodic table, the first thing that you may notice is the number that lies above the symbol. Ingesting or inhaling calcium metal can be fatal due to the burns it can produce.\) Skin contact with calcium metal can cause irritation, corrosion, and chemical burns. Calcium metal reacts exothermically with water or acid.People report is as contributing a mineral, sour, or salty flavor. People and other animals can often taste the calcium ion.Calcium metal is soft enough to cut with a knife, although somewhat harder than the metal lead.Calcium salts are used to produce orange in fireworks. Calcium is used in fireworks to deepen the color. 
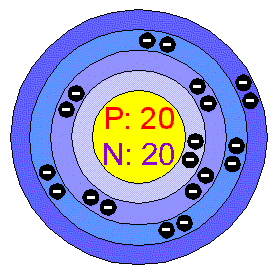
Calcium burns with a dark red color in a flame test.Approximately one third of the mass of the human body is calcium after all water is removed. Calcium is the fifth most abundant element in the human body. 
However, too much calcium can lead to kidney stones or artery calcification. Calcium is the main component of teeth and bones.The top three countries that produce calcium are China, United States and India.Some of the most common compounds found on Earth are limestone (calcium carbonate - CaCO 3), gypsum (calcium sulfate - CaSO 4♲H 2O), fluorite (calcium fluoride - CaF 2) and apatite (calcium fluorophosphate - CaFO 3P or calcium chlorophosphate - CaClO 3P) Calcium is not found free in nature, but calcium compounds are common.Calcium is the 5th most abundant element in the Earth's crust, making up 3.22% of the earth, air, and oceans.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
